Nanotubes
The carbon nanotube, a cylindrical tube of carbon atoms that is sized on the order of 10^-9, has begun to pave the way towards carbon polymers that are lighter and stronger than ever before and have unique, valuable chemical and physical properties. Nanotubes consist of carbon atoms bonded to one another in repeating hexagonal patterns to create a hollow cylinder; the strength of these carbon bonds and the predictable structure of the tubes result in a variety of fascinating chemical and physical properties that will be discussed shortly.
First, though, we must discuss two major types of nanotubes: single-walled (SWNTs) and multi-walled (MWNTs).
First, though, we must discuss two major types of nanotubes: single-walled (SWNTs) and multi-walled (MWNTs).
|
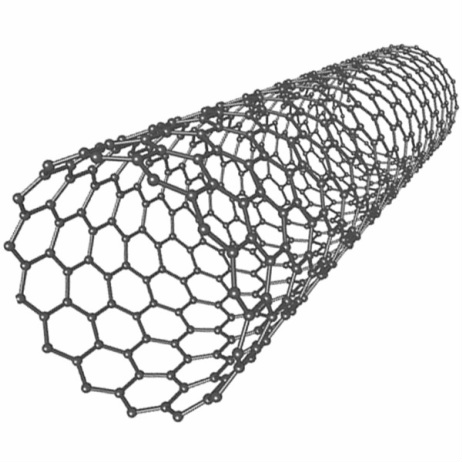
This image is of a single-walled nanotube; note the hexagonal patterns formed by the bonds between the ball-shaped carbon atoms.
|
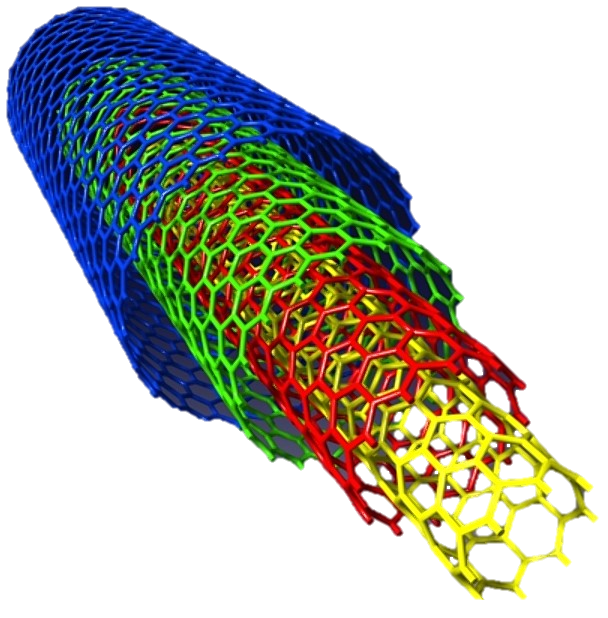
A multi-walled tube, such as the kind shown above, is made up of many concentric single-walled tubes. Because of their additional layers, multi-walled tubes are stronger than the single-walled variety. MWNTs are the favored variety of nanotubes for use in composite materials.
|
PROPERTIES OF NANOTUBES
Carbon nanotubes exhibit chemical and physical properties that make them commercially valuable and interesting. One such property is a high electrical conductivity, which makes the tubes useful for electrical and electrostatic purposes. For example, this conductivity can facilitate electrostatic painting in the automotive industry. By grounding a conductive material, such as a composite material that includes nanotubes, to give it a positive charge before spraying it with negatively-charged paint particles, manufacturers can evenly distribute paint across its surface and, in reducing the amount of paint needed, save money.
Nanotubes are also lightweight and have a high tensile strength, with the result that they function as a strong, useful material. Scientists have measured the tensile strength of multi-walled carbon nanotubes to be as high as 0.15 TPa, which is incredibly strong. The tubes' fantastic tensile strength and low density result in the highest specific strength, or force at failure divided by density, of any known material
Carbon nanotubes exhibit chemical and physical properties that make them commercially valuable and interesting. One such property is a high electrical conductivity, which makes the tubes useful for electrical and electrostatic purposes. For example, this conductivity can facilitate electrostatic painting in the automotive industry. By grounding a conductive material, such as a composite material that includes nanotubes, to give it a positive charge before spraying it with negatively-charged paint particles, manufacturers can evenly distribute paint across its surface and, in reducing the amount of paint needed, save money.
Nanotubes are also lightweight and have a high tensile strength, with the result that they function as a strong, useful material. Scientists have measured the tensile strength of multi-walled carbon nanotubes to be as high as 0.15 TPa, which is incredibly strong. The tubes' fantastic tensile strength and low density result in the highest specific strength, or force at failure divided by density, of any known material
Another interesting property of the tubes is their good thermal conductivity, which allows them to be used for heat transfer in polymers and other materials. MWNTs’ thermal conductivity at room temperature is over than 3000 W/K m, which compares very favorably to metals long considered to be good conductors of heat.
NANOTUBE PRODUCTION
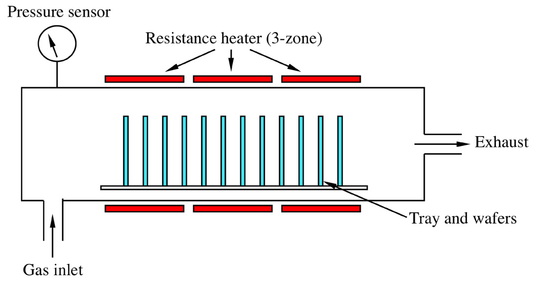
The most common way of producing nanotubes industrially is the Carbon Vapor Deposition, or CVD, process, in which carbon-bearing gases deposit carbon atoms on a growing nanotube. As the nanotube grows longer, its structure stays consistent.
NANOTUBE PRODUCTION
The most common way of producing nanotubes industrially is the Carbon Vapor Deposition, or CVD, process, in which carbon-bearing gases deposit carbon atoms on a growing nanotube. As the nanotube grows longer, its structure stays consistent.
This CVD process requires a catalytic surface on which the tubes can begin to grow. Since industrial producers have traditionally used metal catalysts to provide this necessary surface, the presence of metals has been known to produce some metallic impurities in the final product. These impurities have a negative effect on the stability of the resultant nanotubes.
Some scientists have worked to develop a non-metal catalyst for the CVD process; such a catalyst would remove all possibility of metallic impurities and make for stronger, even more stable nanotubes. As it stands today, 95% purity is the standard industrial grade for MWNTs.
Some specializations are possible during production as well. For example, manufacturers can adjust the size of the nanotubes they make in order to match consumer demand. Beyond size specification, another very interesting alteration during nanotube production is the addition of functional groups, such as hydrogen-bonding sites or animo groups, to the product. These functionalized nanotubes interact in specialized ways with certain other substances and can create additional stability or reactivity as a result.
Nanotubes have been employed for their electrical and thermal conductivity, chemical stability, and incredible strength. Some manufacturers have included them as a high-strength filler in composite materials as a replacement to, or in combination with, traditional fillers such as carbon fibers. In order to understand the advantages of combining the two materials, we now turn to a short description of traditional carbon fibers. On the page "Carbon Fibers and Nanotubes Combined: Multiscale Hybrids," you can find an analysis of the combination of these two filler materials and its impacts on composite materials.
Carbon Fibers
About a thousand times larger than nanotubes, carbon fibers exist at the microscale of 10^-6 meters. The fibers are made up of interlocking sheets of graphene, which in turn are made up of carbon atoms.
PROPERTIES OF CARBON FIBERS
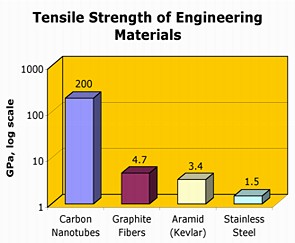
Like nanotubes, the fibers have a notably high tensile strength and are very stiff. See the graph above, which compares the tensile strengths of different engineering materials, to see where fibers fall relative to such materials as stainless steel and nanotubes. The latter two of the three images below depict carbon fiber strands that have been woven together to create a macroscopic sheet of the material.
One major use of carbon fibers is their inclusion in composite materials in order to lend strength and stability to the product. Since nanotubes' inclusion in composite materials is a focus of this website, we will focus our attention on carbon fibers' use in this area.
CARBON FIBERS IN COMPOSITE MATERIALS
To create a carbon fiber composite material (formally referred to as a Carbon-fiber-reinforced polymer), manufacturers take a polymer material such as epoxy resin, known as a matrix, and introduce carbon fibers, known as the reinforcement, into the mix. The resulting material is stronger and stiffer than the polymer alone would have been and is known as a fiber-reinforced polymer. However, the physical makeup of these polymers is not ideal. Interactions between the matrix and the reinforcement are the key to the strength of a composite material, and though carbon-fiber-reinforced polymers are very strong, they are not as strong as they could be; the interactions between the carbon fibers and the matrix are weak. The goal of multiscale hybrids, the focus of the next page on this website, is to use nanotubes as a kind of "binding agent" between the carbon fibers and composite matrix, in order to improve physical interactions between the two materials and thereby increase strength.
SOURCES:
Bierdel, M., Buchholz, S., Michele, V., Mleczko, L., Rudolf, R., Voetz, M., & Wolf, A. (2007, September). Industrial Production of Multiwalled Carbon Nanotubes. Phys. Stat. Sol. 244(11), 3939-3943. doi: 0.1002/pssb.200776101
Demczyka B.G., Wanga Y.M., Cumingsa, J., Hetmana, M., Hana, W., Zettla, A., & Ritchieb, R.O. (2002, September). Direct mechanical measurement of the tensile strength and elastic modulus of multiwalled carbon nanotubes. Materials Science and Engineering, Volume 334(1-2), 173-178. doi: 10.1016/S0921-5093(01)01807-X
Grebler, S., Fries, R., & Simkó, M. (2012, February). Carbon Nanotubes – Part I: Introduction, Production, Areas of Application. NanoTrust Dossiers, 22, 1-5. Retrieved from http://www.musiklexikon.ac.at:8000/ita/nanotrust-dossiers/dossier022en.pdf
Inglefield Jr., D. L., & Long, T. E. (2012). Surface Functionalization of Multi-Walled Carbon Nanotubes with Hydrogen Bonding Functionality for Tailored Polyurethane Nanocomposites. Retrieved from http://www.speautomotive.com/SPEA_CD/SPEA2012/pdf/NN/NN2.pdf
Kim, P., Shi, L., Majumdar, A. & P. L. McEuen (2001, October). Thermal Transport Measurements of Individual Multiwalled Nanotubes. Phys. Rev. Lett., 87. doi: http://dx.doi.org/10.1103/PhysRevLett.87.215502
Li, C., Schäffer, A., Séquaris, J., László, K., Tóth, A., Tombácz, E., Vereecken, H., Ji, R., & Klumpp, E (2012, July). Surface-associated metal catalyst enhances the sorption of perfluorooctanoic acid to multi-walled carbon nanotubes. Journal of Colloid and Interface Science, 377(1), 342-346. doi: 10.1016/j.jcis.2012.03.038
Sharma, S. P. and Lakkad, S. C. (2011, January) Effect of CNTs growth on carbon fibers on the tensile strength of CNTs grown carbon fiber-reinforced polymer matrix composites. Composites Part A: Applied Science and Manufacturing 42(1), 8-15. Retrieved from: http://ac.els-cdn.com.libezp.lib.lsu.edu/S1359835X10002563/1-s2.0-S1359835X10002563-main.pdf?_tid=48ca3b88-10ba-11e4-a31e-00000aacb360&acdnat=1405935480_2f30be48d6dd6337802cb3ff284e344a
Xu, X., Huang, S., Hu, Y., Lu, J., & Yang, Z. (2012, March). Continuous synthesis of carbon nanotubes using a metal-free catalyst by CVD. Materials Chemistry and Physics, 133(1), 95-102. http://dx.doi.org.libezp.lib.lsu.edu/10.1016/j.matchemphys.2011.12.059